Share optimized solutions, professional valve knowledge and industry news
Please enter the relevant terms or keywords you need to consult, and relevant articles will appear in the search results. If you can’t find the answer you need, please feel free to contact us and we will be happy to help. Or you can directly send an email to beauty@shefmon.com
How Are Beauty Treatment Devices Imported from the United States?
- admin
Introduction
Importing beauty treatment devices from the United States has become a popular option for distributors, clinics, salons, and beauty entrepreneurs seeking advanced, high-quality technology. U.S.-made equipment is well known for reliability, strong research foundations, and strict compliance standards.
But importing beauty equipment is not as simple as ordering consumer goods. There are regulations, documentation, customs procedures, and safety standards you must follow to ensure your shipment clears without delays.
This guide walks you step-by-step through how beauty treatment devices are imported from the United States—so you can avoid costly mistakes and ensure smooth delivery.
Legal Requirements Before Importing
Importing beauty treatment devices typically requires compliance with several U.S. and destination-country regulations.
FDA Regulations
Most professional beauty devices that affect the body—such as laser machines, RF devices, IPL systems, and micro-needling tools—are classified as medical devices in the U.S.
This means the manufacturer must follow FDA standards and provide proper documentation.
Device Classification Categories
The FDA divides devices into three categories:
Class I: Low-risk devices (skin scrubbers, simple LED devices)
Class II: Moderate-risk (IPL, RF, laser therapy devices)
Class III: High-risk (rare in beauty industry)
Most beauty machines fall under Class II, meaning more documentation is required.
Checking Device Approval and Compliance
Before importing, request:
FDA registration number
510(k) clearance (if required)
U.S. agent details
Product testing reports
Never import devices without verifying these details, as customs may reject them.
Essential Certifications for Importing
FDA 510(k)
Many Class II devices require FDA 510(k) clearance confirming they are safe and substantially equivalent to an existing approved device.
IEC Safety Certifications
Beauty machines typically require:
IEC 60601 (electrical safety)
EMC testing (electromagnetic compatibility)
These ensure devices meet international safety standards.
Manufacturer Compliance Documents
Suppliers should provide:
Declaration of conformity
Technical specifications
Quality system certifications (like ISO13485)
Finding a Reliable U.S. Supplier
How to Evaluate a Manufacturer
Check:
Years in business
Certifications
Factory audits
Customer reviews
Warranty terms
Requesting Product Documentation
Before purchasing, always request:
Complete FDA documentation
Safety test reports
MSDS (if applicable)
User manuals
Avoiding Scams and Fake Devices
Be cautious of:
Manufacturers without FDA listings
Suspiciously low prices
Unregistered companies
Import Procedures Step-by-Step
Step 1 – Identify the Correct HS Code
Beauty devices fall under codes like:
9018 (medical devices)
8543 (electrical machines)
Correct classification prevents customs delays.
Step 2 – Calculate Duties and Taxes
Import taxes depend on your country and HS code. Use an online tariff calculator or ask your freight forwarder.
Step 3 – Choose the Right Shipping Method
Air freight and express shipping are common. Sea freight is suitable for bulk orders.
Step 4 – Customs Declaration
Importers must submit:
Invoice
Packing list
Bill of lading
Certificates
FDA documents (if required)
Step 5 – Receiving and Inspecting the Goods
Inspect for damage, missing parts, or compliance issues. Report immediately if something is wrong.
Shipping Options from the U.S.
Air Freight
Great for fast shipments but more expensive.
Sea Freight
Cost-effective for large machines but slow.
Express Courier
FedEx, UPS, and DHL offer door-to-door delivery, often the easiest option for smaller devices.
Costs Involved in Importing Beauty Devices
Your total cost includes:
Purchase Cost
Depends on brand, model, and technology.
Shipping Fees
Based on weight, size, and method.
Customs Duties
Import tax varies by country.
Warehouse and Handling Fees
You may incur terminal fees, inspection fees, or storage charges.
Common Challenges and How to Avoid Them
Improper Documentation
Missing FDA or IEC certificates commonly delay shipments.
FDA Compliance Issues
Customs may hold or destroy non-compliant machines.
Customs Delays
Most delays happen due to incorrect HS codes or unclear invoices.
Tips to Ensure Smooth Importation
Work With an Experienced Freight Forwarder
They assist with all paperwork and reduce risk.
Verify Certifications Early
Do not wait until after purchasing the machine.
Maintain Clear Communication With Suppliers
Regular updates prevent misunderstandings and delays.
Why Many Companies Prefer Wholesale from China Instead
Cost Differences
U.S. beauty machines are typically 2–5 times more expensive.
Easier Compliance
Chinese manufacturers usually offer complete export certifications upfront.
Faster Delivery and OEM Options
Factories in China provide white-labeling, custom housing, branding, and marketing support.
Recommendation: SHEFMON Beauty Equipment
If you want professional beauty devices with FDA, CE, ISO certifications, customized branding, and competitive prices, SHEFMON is a strong option.
Popular categories include:
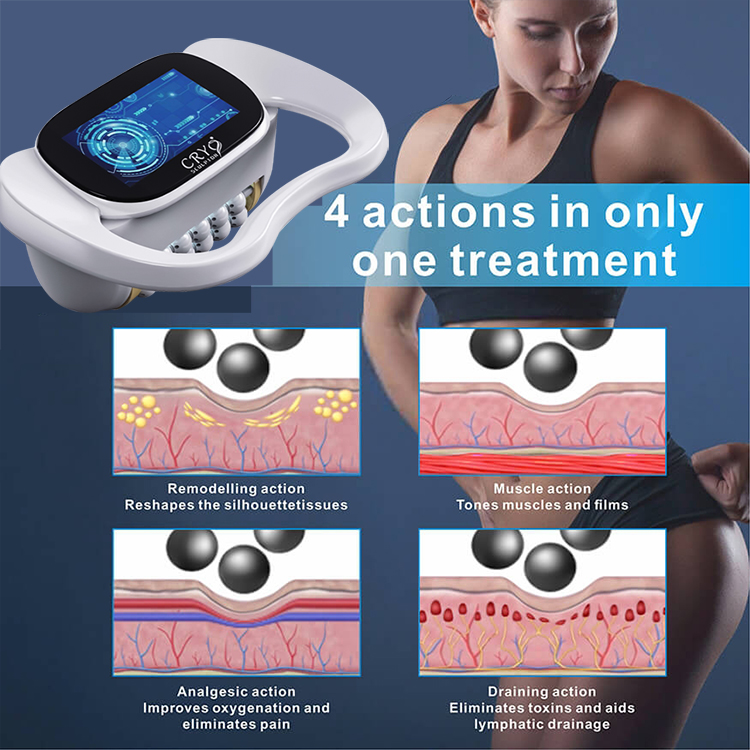
Cryolipolysis
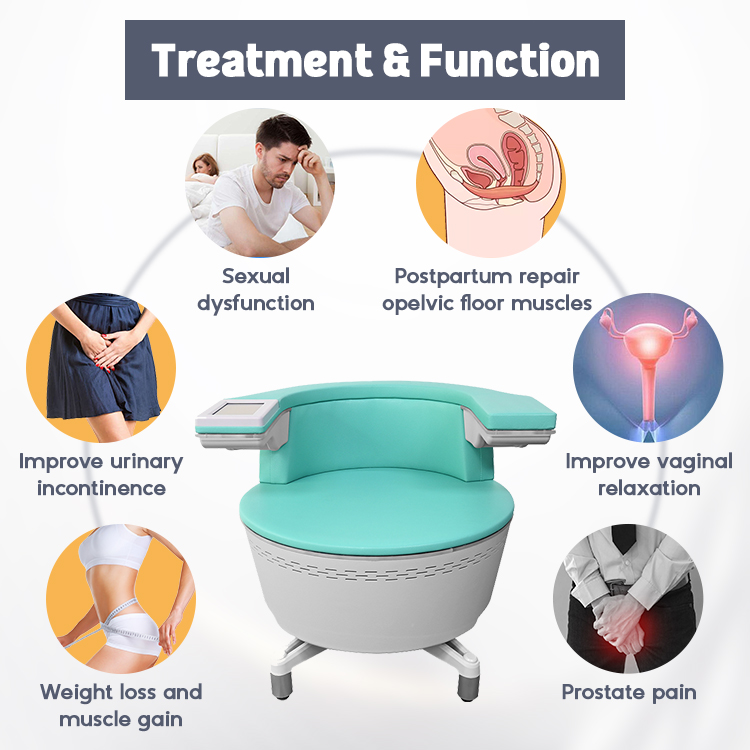
Emsculpt & Pelvic Floor Chairs
HIFU
Hydra facial machines
Laser IPL
RF Cavitation
Shockwave therapy
Visit: https://shefmon.com/
Conclusion
Importing beauty treatment devices from the U.S. involves careful planning, regulatory understanding, and thorough documentation. By verifying FDA compliance, choosing reliable suppliers, and working with experienced freight forwarders, you can ensure your devices pass customs smoothly.
However, many businesses choose Chinese manufacturers like SHEFMON due to lower costs, faster delivery, and easier export documentation. Ultimately, your import path depends on your business goals, budget, and market demands.
FAQs
1. Are all beauty devices from the U.S. FDA-approved?
No. You should verify FDA registration and 510(k) clearance before importing.
2. How long does shipping from the U.S. take?
Air freight takes 3–10 days; sea freight can take 20–45 days.
3. Do I need a special license to import beauty devices?
Some countries require importer registration for medical devices.
4. What happens if my shipment fails FDA inspection?
It may be returned, destroyed, or held indefinitely.
5. Is importing from China easier than from the U.S.?
For many importers—yes. Chinese suppliers often provide complete documentation and lower pricing.